BEST QUALITY FOR YOU
Where Quality Meets Comfort.
PRICE MATCH GURANTEE
Find a better price and we will beat it.
SERVICE YOU CAN TRUST
Local techs and free lifetime phone support


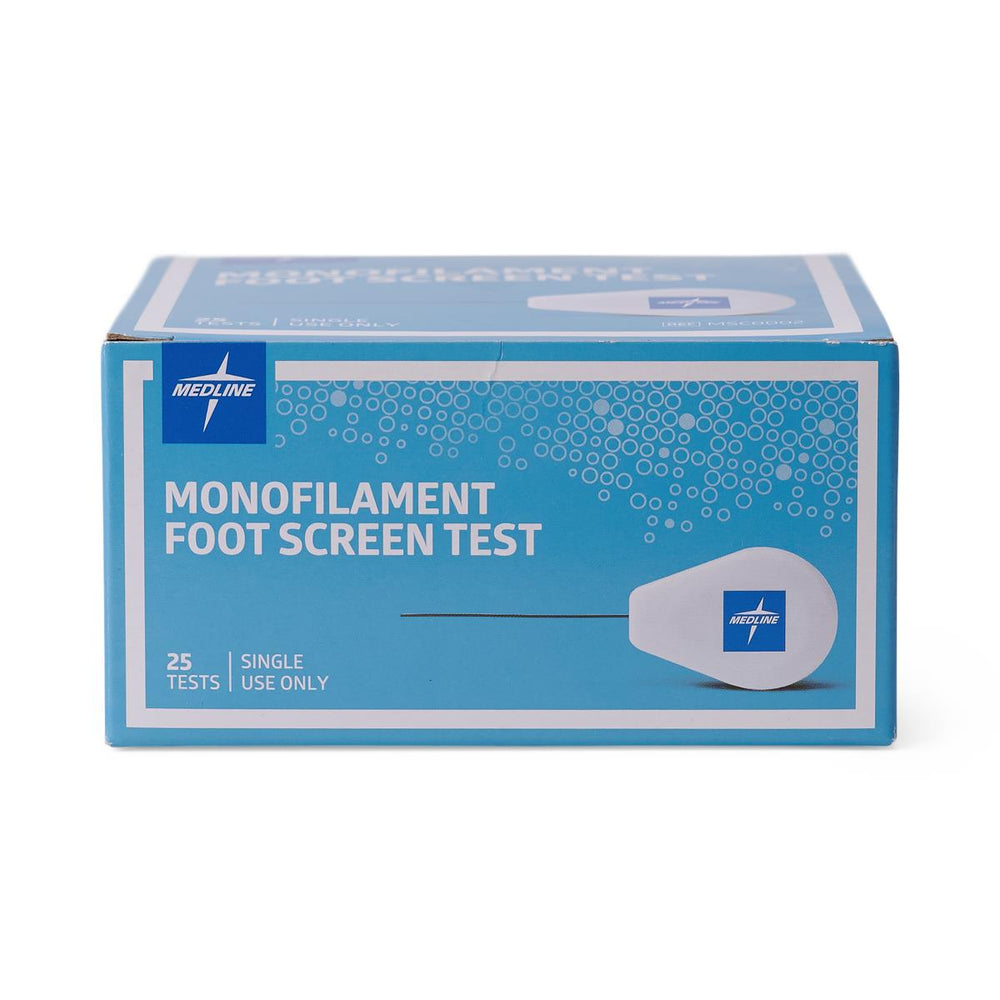









Diabetic Neuropathy Monofilament Testing Device - 10g Semmes-Weinstein Filament for Foot Screening
- $4.68
$4.91- $4.68
- Unit price
- per

Vendor: Medline
Diabetic Neuropathy Monofilament Testing Device - 10g Semmes-Weinstein Filament for Foot Screening







Vendor: Medline
Diabetic Neuropathy Monofilament Testing Device - 10g Semmes-Weinstein Filament for Foot Screening
Description
xProfessional Diabetic Neuropathy Screening Tool for Early Ulcer Prevention
This precision-calibrated monofilament testing device is an essential diagnostic tool for identifying patients at risk of developing diabetic foot ulcers, one of the most serious and costly complications of diabetes mellitus. Designed for use by endocrinologists, podiatrists, primary care physicians, diabetes educators, wound care specialists, and home healthcare providers, this latex-free monofilament delivers standardized 10-gram force testing to assess protective sensation in diabetic patients' feet—enabling early detection of peripheral neuropathy before devastating ulceration, infection, and potential amputation occur.
Clinical Significance & Diagnostic Value
- Early Neuropathy Detection: Identifies loss of protective sensation (LOPS) in diabetic patients before visible tissue damage occurs, allowing for timely intervention with preventive foot care protocols, specialized footwear, and patient education to reduce ulcer risk by up to 60%
- Evidence-Based Screening Standard: Delivers consistent 10-gram force application following Semmes-Weinstein monofilament testing methodology—the gold standard recommended by the American Diabetes Association, International Working Group on the Diabetic Foot, and clinical practice guidelines worldwide
- Cost-Effective Risk Stratification: Provides reliable, reproducible testing at minimal cost compared to complex neurological assessments, enabling systematic screening of large diabetic patient populations in primary care, endocrinology clinics, and community health settings
- Quantifiable Clinical Documentation: Generates objective, measurable data for patient medical records, supporting clinical decision-making, treatment planning, insurance reimbursement, and longitudinal tracking of neuropathy progression over time
- Latex-Free Construction: Safe for patients and healthcare providers with latex allergies or sensitivities, meeting modern clinical safety standards and reducing allergic reaction risks during routine diabetic foot examinations
- Portable & Convenient: Compact, lightweight design fits easily in white coat pockets, medical bags, or examination room drawers, ensuring accessibility for point-of-care testing during routine diabetic follow-up appointments
- Durable Medical-Grade Materials: Engineered for repeated clinical use with consistent force calibration, maintaining diagnostic accuracy through hundreds of patient examinations when used according to manufacturer guidelines
Clinical Applications & Patient Populations
Essential for comprehensive diabetic foot screening programs in patients with Type 1 diabetes, Type 2 diabetes, gestational diabetes with long-term complications, prediabetes with existing neuropathy symptoms, and any diabetic patient with risk factors including poor glycemic control, diabetes duration exceeding 5 years, history of foot ulcers, peripheral vascular disease, visual impairment, renal disease, or advanced age. Particularly critical for high-risk populations including elderly diabetics, patients with HbA1c levels above 7%, individuals with previous amputations, and those with concurrent cardiovascular disease.
Product Specifications
- Force Calibration: 10-gram Semmes-Weinstein monofilament (5.07 gauge)
- Testing Standard: Follows ADA and IWGDF neuropathy screening protocols
- Material: Medical-grade nylon monofilament with ergonomic handle
- Latex Content: Latex-free for allergy safety
- Sterilization: Single-patient use recommended; can be cleaned between uses per facility protocols
- Durability: Maintains calibration through extended clinical use
- Portability: Compact design for point-of-care convenience
- SKU: MEMSC0002
- MPN: MSC0002
- GTIN: 10080196395958
- Unit of Measure: Each (individual testing device)
Evidence-Based Testing Protocol
The monofilament test is performed by applying the filament perpendicular to the skin surface at standardized foot locations (plantar aspects of the great toe, first metatarsal head, third metatarsal head, and fifth metatarsal head) until the filament bends. Patients unable to perceive the 10-gram force at two or more sites are classified as having loss of protective sensation, indicating significant neuropathy and elevated ulcer risk requiring intensive preventive interventions including therapeutic footwear, frequent foot inspections, and specialized podiatric care.
Preventive Care Impact & Outcomes
Systematic monofilament screening programs have demonstrated remarkable success in reducing diabetic foot complications. Studies show that regular neuropathy screening combined with patient education and preventive foot care reduces ulcer incidence by 50-60% and amputation rates by up to 85%. Early identification of at-risk patients enables implementation of protective strategies including custom orthotics, pressure-relieving footwear, intensive patient education about daily foot inspection, prompt treatment of minor injuries, and regular podiatric follow-up—interventions that dramatically improve quality of life and reduce healthcare costs associated with advanced diabetic foot disease.
Healthcare Provider Benefits
For busy clinical practices, this monofilament testing device enables rapid, efficient neuropathy screening that can be completed in under 2 minutes during routine diabetic follow-up visits. The objective, reproducible results support quality metrics reporting, diabetes care accreditation standards (NCQA, HEDIS), and value-based care initiatives that reward preventive screening. Documentation of monofilament testing satisfies Medicare and insurance requirements for comprehensive diabetic foot examinations, supporting appropriate reimbursement for preventive care services.
Procurement & Clinical Program Implementation
Essential equipment for endocrinology practices, primary care clinics, podiatry offices, diabetes education centers, wound care clinics, hospital diabetes programs, community health centers, and home healthcare agencies serving diabetic populations. Cost-effective solution for establishing systematic foot screening protocols that meet clinical practice guidelines and quality improvement standards. Compatible with electronic health record documentation systems and diabetes registry tracking programs for population health management.
Patient Education Integration
The monofilament test provides a powerful teaching moment for patient engagement in diabetic foot care. When patients experience diminished sensation during testing, it creates immediate awareness of their neuropathy risk and motivates adherence to preventive behaviors including daily foot inspections, proper footwear selection, prompt reporting of foot injuries, and regular podiatric care. Healthcare providers can use test results to reinforce the critical connection between glycemic control and neuropathy prevention, supporting broader diabetes self-management goals.
Quality Assurance & Compliance
Meets clinical standards for diabetic neuropathy screening established by the American Diabetes Association, American Podiatric Medical Association, and International Working Group on the Diabetic Foot. Supports compliance with diabetes quality measures including HEDIS Comprehensive Diabetes Care metrics, Medicare Advantage Star Ratings, and Patient-Centered Medical Home recognition criteria that require documented annual foot examinations for diabetic patients.
Shipping & Return
xShipping Policy
We provide shipping worldwide and most of our orders are delivered within 7-15 days.
Return Policy
We have a 30-day return policy, which means you have 30 days after receiving your item to request a return.
To be eligible for a return, your item must be in the same condition that you received it, unworn or unused, with tags, and in its original packaging. You’ll also need the receipt or proof of purchase.
To start a return, you can contact us at cetabo.contact@gmail.com. Please note that returns will need to be sent to the following address: 2236 W Holcombe Blvd, Houston Texas 77030, United States
Related Products
Recently Viewed Products
- Choosing a selection results in a full page refresh.







